New
Image
21.05.2026
Diabetes Scotland sends open letter to Scottish Government over hybrid...
Diabetes Scotland have sent an open letter to the Scottish Government requesting they reconsider the deci...
customer support
customer support
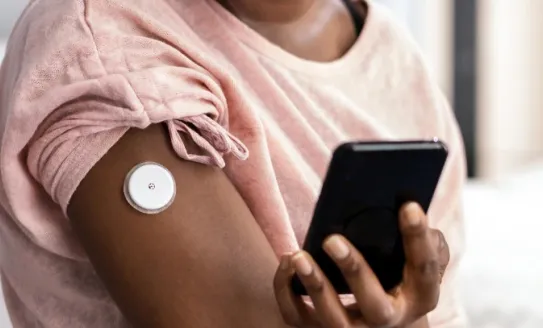
It was our professional conference in April. Read about an important step towards improving the future success of beta cell replacement treatments, and click below to read how continuous glucose monitoring could transform lives for people with type 2 diabetes
Read more
