New
Image
17.03.2026
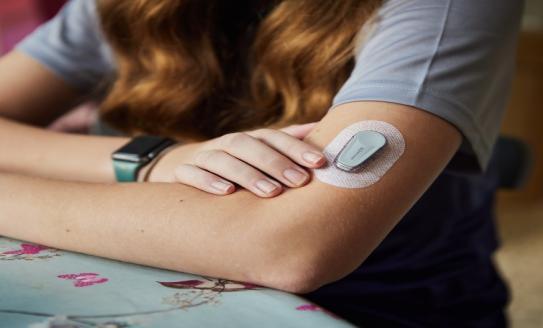
Dexcom G6 CGM being upgraded to Dexcom G7 - what you need to know
Dexcom are phasing out their Dexcom G6 continuous glucose monitor (CGM) and replacing it with the Dexcom ...
customer support
customer support
From research findings supporting SGLT-2 inhibitors as a first-choice type 2 treatment to a real-world study strengthening evidence for teplizumab in early type 1, here are some of the diabetes research developments announced in February 2026.
Read more